Abstract
Background: A substantial number of drugs are being developed from plants for the treatment of various diseases. Lannea egregia (LE) is a woody perennial plant used traditionally in the management of skin disorders and wounds.
Aim: To investigate the biological activities of different solvent extracts of LE bark.
Settings: Fresh stem bark of Lannea egregia was collected from Itabo Lanlate, Oyo State, Nigeria. The authentication was done in Forest Herbarium, Ibadan (FHI), Nigeria. The biological activities of the air-dried sample were carried out in the Department of Pharmacognosy, University of Ibadan, Nigeria.
Materials and methods: Powdered LE bark sample was extracted by gradient extraction. Phytochemical screening was performed on the extracts using standard procedure. In vitro antimicrobial study was performed on 14 strains of bacteria and 5 fungal strains at a concentration range of 25, 50, 75 and 100 mg/mL of the extract using ciprofloxacin and itraconazole as standard. The minimum inhibitory concentration (MIC), minimum bactericidal concentration (MBC) and minimum fungicidal concentration (MFC) were determined. The 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging activity and total phenolic content (TPC) of two active extracts were determined spectrophotometrically.
Results: The phytochemical screening of LE revealed the presence of tannins, terpenoids, flavonoids, anthraquinones, saponins and alkaloids. The dichloromethane (DCM) extract exhibited the highest activity against all the bacterial strains as well as four of the fungal strains. The zones of inhibition (ZI) of bacteria ranged from 9.0 ± 2.0 to 24.6 ± 2.4 mm, MIC of 0.0008 to 12.5 mg/mL and MBC of 25 to 75 mg/mL, while ZI of the fungal strains ranged from 10.3 ± 4.6 to 18.0 ± 5.3 mm, MIC 0.391 – 0.781 and MFC of 50 mg/mL in all the strains. The TPC values of DCM and ethyl acetate (EtOAc) extracts were 1582.47 ± 6.69 and 1579.89 ± 12.77 µg GAE/mg and inhibitory antioxidant activity of 2.54 ± 0.58 and 2.44 ± 0.54, respectively.
Conclusion: These findings provide scientific evidence to support the ethnomedicinal use of Lannea egregia bark for treating skin disorders and wounds.
Introduction
Throughout the development of civilisations, the relationship between human and plants has been very close. Human beings started its civilisation from the forest, which houses diverse fauna and flora species. Traditional medicines provide a reservoir of plant materials that should be tapped. There is often a correlation between local uses of a plant and its activities in biological screening systems (Anokbongoo 1992). Nowadays, a substantial number of drugs being are developed from plants for the treatment of various diseases (Fabricant & Farnsworth 2001).
Ogundeko et al. (2015) and Aiyegoro, Akinpelu and Okoh (2007) reported antibacterial and relaxant activities of Erythrophleum suaveolens (Guill. & Perr.) Brenan (Fabaceae) stem bark; Akinnibosun and Ogedegbe (2009) also worked on the antibacterial potential of Boerhavia diffusa L. (Nyctaginaceae) whole plant; and Falodun et al. (2009) studied the anti-inflammatory activity of Khaya grandifoliola C.DC. (Meliaceae) stem bark, among others.
The medicinal value of plants lies in some chemical substances that produce a definite physiologic action in the human body. These phyto compounds are tannins, terpenoids, alkaloids and flavonoids, which have been found to be very effective in the treatment of various ailments. The phytochemical research based on ethno-pharmacological information is generally considered as an effective approach in the discovery of new anti-infective agents from higher plants (Duraipandiyan, Ayyanar & Ignacimuthu 2006).
Chatterjee and Fleck (2011) established that antimicrobial resistance is a global problem that affects all countries as many bacterial and fungal infections are widespread worldwide. The emergence of resistance to numerous antibiotics necessitates a search for new drugs. The goal of the antimicrobial therapy is to effectively eradicate pathogenic organisms while minimising drug toxicities. Various factors affect the treatment outcomes of infectious diseases, including host defence mechanisms, sites of infection, virulence of the pathogens and pharmacologic properties of the antimicrobial agents used to treat infections (Kingston 2008).
Clinically useful antibiotics now in use have major side effects, such as being neurotoxic, nephrotoxic, ototoxic or hypertensive, and few others cause severe damage to the liver, as well as bone marrow depression, aside from the narrow spectrum of the antimicrobial activity as asserted by Aiyegoro and Okoh (2009). The World Health Organization (2014) also reported that infectious pathogens have developed resistance to almost all known antibiotics. However, abundant medicinal plants have been used in many forms over the years to treat, manage or control human ailments (Prescott, Harley & Klein 2002). Therefore, any effort to further explore medicinal or natural products from botanical flora towards improving healthcare deliveries deserves attention.
Lannea egregia Engl. and K. Krause (Anacardiaceae), like other Lannea species, had been used in folk medicines in African countries to treat various ailments in humans. The stem bark is used externally to treat ulcers, sores and leprosy (Burkill 1985). A decoction is drunk against gastric pains, diarrhoea, oedema, paralysis, epilepsy and madness (Jansen 2005). In Nigeria and the Central African Republic, stem bark decoction is drunk as a stomachic to improve the haemoglobin level and as part of vermifuge medicine (Arbonnier 2004). The macerated root is used in a poultice for the treatment of wounds (Neuwinger 2000). The leaves of Lannea egregia are also used traditionally as an anticancer herb (Soladoye et al. 2010). In Burkina Faso, root decoction is used to cure hernia and leaf decoction is used to cure haemorrhoids (Neuwinger 2000).
Lannea egregia is a deciduous tree that grows up to a height of 590.55 in. (15 m), and is spread across the savannas in Guinea to Nigeria (Jansen 2005). In Guinea, Ivory Coast and Benin, this species shares the same local name as L. barteri (Oliv.) Engl. (Arbonnier 2004) and undoubtedly has the same usage. It is commonly known as false marula and locally known as ekudan (KO&S) in Nigeria (Yoruba). Lannea egregia is a promising medicinal plant, and the result of this study is in full agreement with its traditional usage for treating microbial infections (such as Staphylococcus aureus, Candida albicans, etc.) and wounds healing. To the best of our knowledge, this study is the first report on the preliminary phytoconstituents, antimicrobial and free radical inhibitory properties of L. egregia, which had enjoyed traditional methods for treating skin disorders in Southwest Nigeria. There was no previous study on this species; however, antimicrobial, radical scavenging activities and acetylcholinesterase inhibitions, as well as anthelmintic activity of Lannea barteri have been carried out (Koné et al. 2011).
This work was designed to investigate the phytochemical, antimicrobial and antioxidant properties of Lannea egregia Engl. and K. Krause, with the aim of establishing the presence of bioactive constituents responsible for its ethnomedicinal use.
Materials and methods
Plant collection
The fresh stem bark of Lannea egregia was collected in February 2015 from the savannah region of Itabo Lanlate, Oyo State, Nigeria. The plant species was sent to Forest Herbarium, Ibadan (FHI) for identification and authentication. Voucher specimen was kept at the FHI, with the file number 110395.
Preparation of the extract
The air-dried powdered stem bark was successively extracted with n-hexane, dichloromethane (DCM), ethyl acetate (EtOAc) and methanol (MeOH) for 72 h using a fivefold excess of solvent. Filtrates were then concentrated to dryness in vacuo at 30 °C. All the biological activities were carried out in the Department of Pharmacognosy, Faculty of Pharmacy, University of Ibadan, Nigeria.
Preliminary phytochemical screening
Preliminary qualitative chemical tests were carried out on the solvent extracts of Lannea egregia (stem bark) using standard procedures to identify the phytoconstituents (Harborne 1998; Sofowora 2008; Trease & Evans 1989). Secondary metabolites such as alkaloids, flavonoids, tannins, saponins, terpenoids, cardiac glycoside and anthraquinones were determined.
Alkaloids
Each extract (0.01 g) was diluted with 10 mL of 1% HCl, boiled on a water bath and filtered. To 5 mL of the filtrate, 2 mL of diluted ammonia was added. After this, 5 mL of chloroform was added and gently shaken to extract the alkaloidal base. The chloroform layer was extracted with 10 mL of acetic acid. This was divided into two portions. Mayer’s reagent was added to one portion and Draggendorff’s reagent to the other.
The formation of a cream (with Mayer’s reagent) or reddish-brown precipitate (with Draggendorff’s reagent) was regarded as positive for the presence of alkaloids.
Flavonoids (lead acetate test)
To a 5 mg/mL aqueous solution of the extract, 1 mL of 10% lead acetate solution was added. The production of a yellow precipitate was an indication of flavonoids.
Tannins (ferric chloride test)
The extract (0.01 g) was boiled in 10 mL of water in a test tube and then filtered. A few drops of 0.1% ferric chloride were added and observed for brownish green or a blue-black colouration, which signified the presence of tannins.
Saponins (frothing test)
Distilled water (5 mL) was added to 0.01 g of the extracts in test tubes. The solutions were shaken vigorously and observed for a stable persistent froth. The frothing was mixed with three drops of olive oil and shaken vigorously, after which it was observed for the formation of an emulsion.
Anthraquinones (Borntrager’s test)
The extract (0.01 g) was boiled with 10 mL of benzene and filtered while hot. The filtrate was shaken with 5 mL of chloroform. The chloroform layer was pipetted into another test tube, and 1 mL of diluted ammonia was added. The resulting solution was observed for colour changes. Pink, red or violet colour at the ammoniacal phase indicated the presence of anthraquinones.
Terpenoids (Salkowski test)
Chloroform (2 mL) was added to 0.01 g each of the extracts. Concentrated H2S04 (3 mL) was carefully added to form a layer. A reddish-brown colouration of the interface indicated the presence of terpenoids.
Cardiac glycosides (Keller–Killiani test)
Each extract (0.01 g) was dissolved in 2 mL of glacial acetic acid containing one drop of ferric chloride solution and was diluted with 5 mL of water. They were underlayed with 1 mL of concentrated sulphuric acid. A brown ring at the interface indicated the presence of a deoxysugar characteristic of cardenolides.
Antibacterial test
Bacteria strains used
Plant extracts were screened against 14 clinical strains of bacteria obtained from the Department of Pharmaceutical Microbiology of Olabisi Onabanjo University (OOU), Ago-iwoye, Ogun-State, Nigeria, in September 2015. These included six Gram-negative strains – Klebsiella pneumonia, Proteus mirabilis, Escherichia coli, Pseudomonas aeruginosa, Shigella dysenteriae and Salmonella typhi – and eight Gram-positive strains: Staphylococcus aureus, Bacillus subtilis, Staphylococcus epidermidis, Bacillus cereus, Enterococcus faecalis, Bacillus aureus, Bacillus coagulans and methicillin-resistant Staphylococcus aureus (MRSA). These microorganisms were grown in nutrient broth.
Antibacterial screening procedure
The antibacterial activities of the extracts were tested against the selected strains using agar well diffusion method as described by Mbata, Debiao and Saikia (2008). An amount of 20 mL of sterilised nutrient agar medium was poured into each sterile Petri dish and allowed to solidify. The test bacteria cultures were standardised to 0.5% McFarland standard (NCCLS 1993) and evenly spread over the appropriate media with the aid of a swab stick.
Then wells of 6 mm were made in the medium using a sterile cork borer. Different concentrations of sample solutions were prepared followed by appropriate dilutions to the required concentrations (25, 50, 75 and 100 mg/mL). These concentrations (at 0.1 mL) were transferred into separate wells, followed by the incubation of the plates at 37 °C for 24 h. After the incubation period, the zones of growth inhibition (ZI) were observed and measured using transparent ruler (Mbata et al. 2006). Each test was repeated three times. The mean of the triplicate tests ± their standard error of mean (SEM) was calculated and recorded as the diameter of zone of inhibition. Standard sensitivity discs of selected antibiotics – Septrin (SXT: 30 µg), Ciprofloxacin (CPX: 10 µg) and Pefloxacin (PEF: 10 µg/mL) – were used as positive controls. Active plant extracts showing visible zones of inhibition were further tested at lower concentrations to determine their minimum inhibitory concentration (MIC), using the broth microdilution method in 96-well microtitre plate. The minimum bactericidal concentration (MBC) was determined by subculture of the preparations that have shown no evidence of growth in the MIC determination assay. These subcultures were made in nutrient agar plates (Grierson & Afolayan 1999).
Antifungal screening procedure
Antifungal activity of all the plant extracts was tested using potato dextrose agar well diffusion method (Mbata et al. 2006) against four clinical isolates of fungal strains: Penicillium species, Aspergillus niger, Candida albicans, Fusarium verticiloides and Aspergillus flavus. About 20 mL of freshly prepared and sterilised potato dextrose agar medium (Lab M limited, Heywood, Lancashire, United Kingdom) was poured into each petri dish and allowed to solidify. The test fungal cultures at an equivalent of 0.5% McFarland standard (NCCLS 1993) were evenly spread over the respective plates, with the aid of swab stick. Then wells of 6 mm were made following the same procedure as done for the antibacterial test, and the plates were incubated at room temperature (25 °C – 28 °C) for 5 days; this was done according to the recommendation of NCCLS M27 – A2 protocol as modified by Hahn and Hamdan (2000) and Hahn et al. (2002). The ZI were determined accordingly. A standard drug (Itraconazole) was used as a positive control.
Determination of minimum bactericidal and fungicidal concentrations
Minimum bactericidal concentration (MBC) and minimum fungicidal concentration (MFC) assays were carried out as described by Cheesbrough (2006). Fresh nutrient agar and potato dextrose agar plates were inoculated with one loopful of culture taken from each of the broth cultures that showed no growth in the MIC tubes. The MBC assay plates were incubated at 37 °C for 48 h, while MFC assay plates were incubated at 25 °C for 72 h. After the incubation period, the lowest concentration of the extract that did not produce any bacterial or fungal growth on the solid medium was regarded as MBC and MFC values for each extract (Irkin & Korukluoglu 2007).
Radical scavenging assays using 2, 2-diphenyl-1-picrylhydrazyl
The 2, 2-diphenyl-1-picrylhydrazyl (DPPH) assay was determined using the electron donating ability of the extracts, which was measured by bleaching a purple solution of DPPH radical with ascorbic acid as standard according to the method described by MacDonald-Wicks, Wood and Garg (2006). The DPPH (0.0035 mg) was prepared to make 0.01 mM of the solution. Extracts (50, 25, 12.5, 6.25, 3.125 and 1.562 µg/mL) were added to 0.5 mL of 0.01 mM DPPH–methanol solution. The concentrations of the extracted stock solution were obtained through multifold serial dilutions. The reaction mixture was incubated in a dark room for 30 min, and the free radical scavenging ability was estimated by measuring the absorbance of the mixtures against a blank at 517 nm with the spectrumlab 752s UV-VIS spectrophotometer. The DPPH radical stock solution was freshly prepared for the reaction, and precautionary measures were taken to reduce the loss of free radical activity during the experiment. The inhibition percentage of DPPH radicals by the extract was calculated using the following formula:
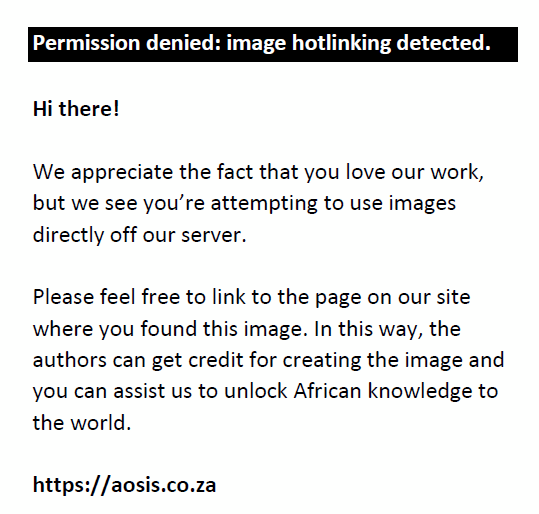
where Absblank = absorbance of the control reaction (all reagents except plant extract):
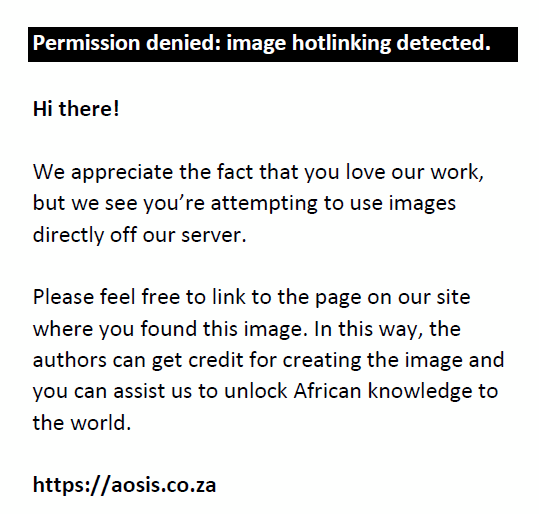
Extract concentration providing 50% inhibition (IC50) was calculated from the graph of inhibition percentage plotted against extract concentration. The L-ascorbic acid was used as positive control. Tests were carried out in triplicate, and the average value was taken.
Estimation of total phenolic content
Total phenolic content (TPC) was determined using the Folin-Ciocalteu method described by Singleton, Orthofer and Lamuela-Raventos (1999), which was based on a colorimetric oxidation and reduction reaction. Folin-Ciocalteu reagent (5 mL) was added to 100 µg/mL aliquots of the extracts. After 3 min, 4 mL of Na2CO3 (7.5%) solution in distilled water was added to the mixture and the content was thoroughly mixed. The mixture was incubated for 30 min after which the absorbance was determined spectrophotometrically at the absorbance of 765 nm using spectrumlab 752s UV-VIS spectrophotometer. A linear dose-response regression curve was generated using absorbance reading of gallic acid at 765 nm wavelength. Results were expressed as µg GAE/mg of the dry weight of the extracts.
Results
Preliminary phytochemical screening result of L. egregia stem bark
The result of the preliminary phytochemical screening of Lannea egregia stem bark extracts using gradient extraction, as shown in Table 1, revealed that cardiac glycoside and saponins were absent in all the extracts except in the DCM where saponins were present. Anthraquinones and terpenoids were present in n-hexane extract, but flavonoids, alkaloids and tannins were absent. However, flavonoids, tannins and terpenoids were found in abundance in the DCM extract, while alkaloids and anthraquinones were also present. Terpenoids and tannins were abundant in the EtOAc extract, while flavonoids, alkaloids and anthraquinones were present. Also, the MeOH extract contained abundant terpenoids and tannins; alkaloids were present but no flavonoids.
| TABLE 1: Preliminary phytochemical screening of L. egregia stem bark extracts. |
Antibacterial screening result of L. egregia stem bark extracts
The antibacterial activities of the extracts were examined in this study. The results are given in Table 2. The values of the zones of growth inhibition (ZI) produced by n-hexane was between 6.0 and 10.0 ± 3.0, DCM ZI values ranged from 9.0 ± 2.0 to 24.6 ± 2.4, EtOAc was between 8.6 ± 2.7 and 20.6 ± 1.2, while MeOH was 11.3 ± 2.7 to 15.3 ± 3.0 mm, respectively. The DCM extract exhibited the highest activity on all the bacterial strains, with ZI ranging from 9.0 ± 2.0 to 24.6 ± 2.4 mm. The negative control plates showed no activity as there was bacteria growth on the plates. This implies that the vehicle solvent did not interfere with the activities shown by the extracts.
| TABLE 2: Inhibition zones (mm) of L. egregia stem bark extracts on test bacteria strains. |
The DCM extract exhibited competing activities with CPX on some of the bacteria strains and was more active than PEF (Figure 1) on most strains. In Proteus mirabilis, the extract at 100 mg/mL had ZI of 23.0 mm, CPX had 22.5 mm, while PEF was not active. Also, the extract was able to inhibit the activity of Pseudomonas aeruginosa at ZI of 23.0 mm, while CPX inhibited the growth at 24.0 mm. It was observed that the activities of the DCM extract were concentration-dependent that is, the higher the concentrations, the higher the activity.
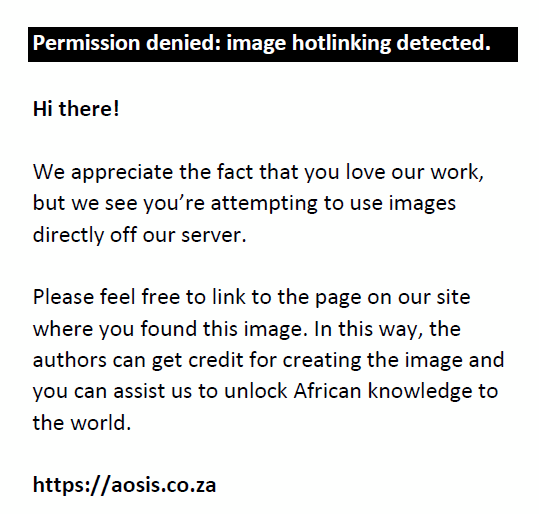 |
FIGURE 1: Competing activities of the most active extract (dichloromethane – DCM) with the standards (CPX – ciprofloxacin, 10 μg/mL: PEF, pefloxacin, 10 μg/mL, MeOH, methanol). |
|
Antifungal screening
The results of antifungal activities of the extracts are presented in Table 3. The DCM extract also showed better activity than others on five strains of fungi: Penicillium species, Candida albicans, Aspergillus niger, Fusarium verticiloides and Aspergillus flavus. The ZI on fungal strains ranged from 10.3 ± 4.6 to 18.0 ± 1.5 mm. The inhibition zones of 6.0 mm indicated little or no activity; inhibition was confined to the well diameter only with no clear ZI. However, the comparable activities of the DCM extract to standard antifungal drug (itraconazole) revealed that the extract exhibited good activity at 100 mg/mL, with better activity observed on Aspergillus flavus than itraconazole. Also, it was revealed that EtOAc extract had better performance on Fusarium verticiloides than DCM and itraconazole.
| TABLE 3: Inhibition zones (mm) of L. egregia stem bark extracts on test fungi strains. |
Minimum inhibitory concentrations and minimum bactericidal/fungicidal concentrations of the dichloromethane extract against different bacterial and fungal strains
The most active extract (DCM) was further tested at lower concentrations to determine the lowest concentration at which it would display activity. The MIC of the DCM extract on bacteria was found to range from 0.0008 mg/mL (Escherichia coli) to 12.5 mg/mL (Enterococcus faecalis), while its lowest inhibitory concentrations on fungi ranged from 0.391 mg/mL to 1.562 mg/mL, as shown in Table 4. The minimal bactericidal and fungicidal concentrations of the extract (Table 4) ranged from 25 mg/mL to 75 mg/mL and 50 mg/mL, respectively.
| TABLE 4: Minimum inhibitory concentrations and minimum bactericidal/fungicidal concentrations of the dichloromethane extract against different bacterial and fungal. |
2, 2-diphenyl-1-picrylhydrazyl free radical scavenging activity of the dichloromethane and ethyl acetate extracts
The two active extracts (DCM and EtOAc) were further subjected to DPPH and TPCs to determine their free radical scavenging activities. The violet colour of DPPH free radical changed to yellow on the addition of these two extracts, which indicated the conversion of DPPH free radicals to DPPH antioxidant compounds. The IC50 values of the extracts are presented in Figure 2. The IC50 of the DCM extract was 2.54, while the value of EtOAc extract was 2.44 and that of ascorbic acid was 5.42. The indication is that the lower the IC50, the higher the activity.
 |
FIGURE 2: IC50 values of dichloromethane and ethyl acetate extracts in 2, 2-diphenyl-1-picrylhydrazyl assay. DCM, dichloromethane; EtOAc, Ethyl acetate. |
|
Total phenolic content of dichloromethane and ethyl acetate extracts of Lannea egregia’s stem bark
The TPC of DCM and EtOAc extracts of L. egregia’s stem bark was measured using the Folin-Ciocalteu method. The results presented in Table 5 show that the TPC values of DCM and EtOAc extracts were 1582.47 ± 6.69 and 1579.89 ± 12.77 µgGAE/mg of extract and inhibitory antioxidant.
| TABLE 5: Total phenolic contents of dichloromethane and ethyl acetate extracts. |
Discussion
The presence of phytochemicals such as flavonoids, alkaloids, terpenoids, tannins and anthraquinones in the stem bark of L. egregia justified its use as a potential medicinal plant in the genus Lannea, which is in line with secondary metabolites found in L. coromandelica and L. alata (Islam et al. 2002; Kumar & Jain 2015; Okoth, Chenia & Koorbanally 2013). The phytochemical profile obtained from this study was similar to that of Kumar and Jain (2015), but differs from their report by the absence of saponins from EtoAC and MeOH extracts.
Havsteen (2010) reported that the presence of flavonoids in plants indicated their significance in pharmaceutical industries for the production of antimicrobial, antioxidant, anticancer and other degenerative diseases drugs.
This validates the result of Soladoye et al. (2010) on the ethnobotanical survey of anticancer plants in Ogun-State, Nigeria, where L. egregia was mentioned as an anticancer plant.
The antimicrobial activity of the DCM extract may be attributed to the abundance of flavonoids in the extract as most compounds isolated from Lannea species are flavonoid compounds. For instance, Islam and Tahara (2000) isolated flavonoids and tannins polyflavonoids (Islam et al. 2002) from Lannea coromandelica. Flavonol was reported to have been isolated from the roots of Lannea alata (Engl.) Engl (Okoth et al. 2013).
Also, Cowan (1999) reported that flavonoids are synthesised by plants to counter microbial infections, which make them effective antimicrobial substances against a wide array of microorganisms.
The antioxidant results of this study revealed that L. egregia stem bark is a good source of natural radical scavenging activity, as DCM and EtOAc extracts were able to decolourise the DPPH and TPC solutions. Sannigrahi et al. (2009) reported that the degree of discolouration is an indication of the free radical scavenging potential of the sample/antioxidant by their hydrogen-donating abilities. These results corroborate the work of Ouattara et al. (2011), which established the antioxidant activities of L. acida, L. velutina and L. microcarpa stem bark and concluded that the activities of these Lannea species as good antioxidant plants were because of the high quantity of TPC (Karon et al. 2007). The lower the inhibitory antioxidant value, the higher the activity is. This study is in correlation with previous studies conducted on Lannea species, which revealed that the genus has good antimicrobial and better free radical scavenging activities. Diallo et al. (2001) and Maiga et al. (2006) established the free radical scavenging and antifungal activities of Lannea velutina against Cladosporium cucumerinum. Also, Queiroz et al. (2003) reported antioxidant and low mutagenic activities of Lannea edulis against the strain TA97 of Salmonella typhimurium. Furthermore, Lannea acida has been reported to possess antibacterial activity against a wide range of bacteria, including MRSA (Koné et al. 2004). Numerous antibacterial and antioxidant compounds have been isolated from Lannea species; Okoth et al. (2013) isolated the arabinofuranoside and rhamnopyranoside from the root of L. alata (Engl.) Engl., which showed antioxidant activity comparable to ascorbic acid. Also, Lanneaflavonol, dihydrolanneaflavonol, myricitrin and betmidin isolated from the root of L. alata exhibited good antibacterial and radical scavenging activity with the glycosides (Okoth et al. 2013). The activities so far from the extracts of L. egregia stem bark support its traditional claims of antimicrobial and wound healing properties.
Conclusion
The stem bark was collected from L. egregia to investigate its antimicrobial and antioxidant activities, with the aim of establishing the presence of bioactive constituents responsible for its ethnomedicinal use. Four organic solvent extracts (n-hexane, DCM, EtOAc and MeOH) were used for the study. The study revealed that the DCM extract showed maximum antimicrobial and better free radical scavenging activities when compared to others.
The results of this study have shown that L. egregia plant is a good source of antimicrobial agents and a potentially good radical scavenger, which justify its traditional use in treating skin disorders, wounds, cancer and some other degenerative diseases. More research should be conducted on the biological activities (such as cancer assay, hypertension and other degenerative diseases assays) of L. egregia stem bark to validate and justify its many traditional uses.
Acknowledgements
The authors are grateful to those who contributed immensely to the success of this study. They appreciate the effort of Mr Ale Oluwatosin of the Department of Pharmaceutical Chemistry, University of Ibadan, and Dr Ibraheem O. Lawal of the Biomedicinal Research Centre, Forestry Research Institute of Nigeria, Ibadan, for their immense contribution towards the completion of this study.
Competing interests
The authors declare that they have no financial or personal relationships which may have inappropriately influenced them in writing this article.
Authors’ contributions
B.O.R. designed the study, coordinated the collection and storage of plant materials, carried out the experiments and put the manuscript together. A.M.S. was responsible for the supervision of plant collection and identification as well as the laboratory experiments, and made substantial contributions to revise the manuscript critically. E.O.A. provided the microorganisms used and took a lead role in carrying out antimicrobial assay. All authors read and approved the final manuscript.
References
Aiyegoro, O.A., Akinpelu, D.A. & Okoh, A.I., 2007, ‘In vitro antibacterial potentials of the stem bark of red water tree (Erythrophleum suaveolens)’, Journal of Biological Sciences 7(7), 1233–1238. https://doi.org/10.3923/jbs.2007.1233.1238
Aiyegoro, O.A. & Okoh, A.I., 2009, ‘Use of bioactive plant products in combination with standard antibiotics: Implications in antimicrobial therapy (a review)’, Journal of Medicinal Plants Research 3(13), 1147–1152.
Akinnibosun, F.I. & Ogedegbe, D.I., 2009, ‘Investigation on the antibacterial activity of the aqueous and ethanolic extracts of the leaves of Boerhavia diffusa L’, Science World Journal 4(2), 15–18.
Anokbongoo, W.W., 1992, ‘The role of African traditional medicine in health care delivery alongside modern medicine, Botany 2000: East and Central Africa’, Natural Products Research Network for Eastern and Central Africa (NAPRECA) Monograph 5, 25–35.
Arbonnier, M., 2004, Trees, shrubs and lianas of West African dry zones, CIRAD, Margraf Publishers Gmbh, MNHN, Paris, France, p. 573.
Burkill, H.M., 1985, The useful plants of west tropical Africa, 2nd edn., vol. 1, Families A–D, Royal Botanic Gardens, Kew, Richmond, United Kingdom, p. 960.
Chatterjee, P. & Fleck, F., 2011, ‘Mobilizing political will to contain antimicrobial resistance’, Bulletin of the World Health Organization 89, 168–169. https://doi.org/10.2471/BLT.11.030311
Cheesbrough, M., 2006, District laboratory practice in tropical countries, 1st edn., Cambridge University Press, Cambridge, UK, p. 434.
Cowan, M.M., 1999, ‘Plant products as antimicrobial agents’, Clinical Microbiology Reviews 12(4), 564–582. https://doi.org/10.1128/CMR.12.4.564
Diallo, D., Marston, A., Terreaux, C., Touré, Y., Paulsen, B.S. & Hostettmann, K., 2001, ‘Screening of Malian medicinal plants for antifungal, larvicidal, molluscicidal, antioxidant and radicals scavenging activities’, Phytotherapy Research 15, 401–406. https://doi.org/10.1002/ptr.738
Duraipandiyan, V., Ayyanar, M. & Ignacimuthu, S., 2006, ‘Ethnomedicine’, BioMedical Central Complementary and Alternative Medicine 635, 6–25.
Fabricant, D.S. & Farnsworth, N.R., 2001, ‘The value of plants used in traditional medicine for drug discovery’, Environmental Health Perspectives 109, 69–75.
Falodun, A., Siraj, R., Quadir, M.I., Khan, T., Sheraz, A. & Choudhary, M.I., 2009, ‘Chemical composition and insecticidal activity of volatile oil of Khaya grandifoliola’, Medicinal and Aromatic Plant Science and Biotechnology 3(Special Issue 1), 61–63.
Grierson, D.S. & Afolayan, A.J., 1999, ‘Antibacterial activity of some indigenous plants used for the treatment of wounds in the Eastern Cape, South Africa’, Journal of Ethnopharmacology 66, 103–106. https://doi.org/10.1016/S0378-8741(98)00202-5
Hahn, R.C. & Hamdan, J.S., 2000, ‘In vitro susceptibilities of Paracoccidiodies brasiliensis yeast form to antifungal drugs’, Mycoses 43, 403–407. https://doi.org/10.1111/j.1439-0507.2000.00606.x
Hahn, R.C., Fontes, C.J.F., Batista, R.D. & Hamdan, J.S., 2002, ‘In vitro comparison of activities of terbinafine & itraconazole against Paracoccidiodies brasiliensis’, Journal of Clinical Microbiology 40, 2828–2831. https://doi.org/10.1128/JCM.40.8.2828-2831.2002
Harborne, J.B., 1998, ‘Phytochemical methods’, 3rd edn., Chapman & Hall, London, p. 5–30.
Havsteen, B.H., 2010, ‘The biochemistry and medical significance of the flavonoids’, Pharmacology & Therapeutics 96(2–3), 67–202.
Irkin, R. & Korukluoglu, M., 2007, ‘Control of Aspergillus niger with garlic, onion and leek extracts’, African Journal of Biotechnology 6, 384–387.
Islam, M.T., Ito, T., Sakasai, M. & Tahara, S., 2002, ‘Zoosporicidal activity of polyflavonoid tannin identified in Lannea coromandelica stem-bark against phytopathogenic Oomycete Aphanomyces Cochlioides’, Journal of Agricultural and Food Chemistry 50, 6697–6703. https://doi.org/10.1021/jf020554g
Islam, Md. T. & Taharaa, S., 2000, ‘Dihydroflavonols from Lannea coromandelica division of applied bioscience’, Phytochemistry 54, 901–907.
Jansen, P.C.M., 2005, ‘Lannea barteri (Oliv.) Engl. [Internet] record from PROTA4U’, in P.C.M. Jansen & D. Cardon (eds.), PROTA 3: Dyes and tannins/colorants et tanins, [CD-Rom], Plant resources of tropical Africa/Ressources végétales de l’Afrique tropicale Wageningen, pp. 345–346.
Karon, A.E., Archer, J.R., Sotir, M.J., Monson, T.A. & Kazmierczak, J.J., 2007, ‘Human multidrug-resistant Salmonella Newport Infections, Wisconsin’, Emerging Infectious Diseases 13(11), 1777–1778.
Kingston, W., 2008, ‘Irish contributions to the origins of antibiotics’, Irish Journal of Medical Science 177(2), 87–92. https://doi.org/10.1007/s11845-008-0139-x
Koné, W.M., Atindehou, K.K., Terreaux, C., Hostettmann, K., Traoré, D. & Dosso, M., 2004, ‘Traditional medicine in north Côte-d’Ivoire: Screening of 50 medicinal plants for antibacterial activity’, Journal of Ethnopharmacology 93, 43–49. https://doi.org/10.1016/j.jep.2004.03.006
Kumar, T. & Jain, V., 2015, ‘Appraisal of total phenol, flavonoid contents and antioxidant potential of folkloric Lannea coromandelica using in vitro and in vivo assays’, Scientifica 2015, Article ID 203679, 13 Pages.
MacDonald-Wicks, L.K., Wood, L.G. & Garg, M.L., 2006, ‘Methodology for the determination of biological antioxidant capacity in Vitro: A review’, Journal of Science of Food and Agriculture 86, 2046–2056. https://doi.org/10.1002/jsfa.2603
Maiga, A., Malterud, K.E., Diallo, D. & Paulsen, B.S., 2006, ‘Antioxidant and 15-lipoxygenase inhibitory activities of the Malian medicinal plants Diospyros abyssinica (Hiern) F. White (Ebenaceae), Lannea velutina A. Rich (Anacardiaceae) and Crossopteryx febrifuga (Afzel) Benth. (Rubiaceae)’, Journal of Ethnopharmacology 104, 132–137. https://doi.org/10.1016/j.jep.2005.08.063
Mbata, T.I., Debiao, L. & Saikia, A., 2008, ‘Antibacterial activity of the crude extract of Chinese Green Tea (Camellia sinensis) on Listeria minocytogenes’, International Journal of Microbiology 7, 1571–1573.
National Committee for Clinical Laboratory Standards (NCCLS), 1993, Dilution antimicrobial susceptibility for bacteria that group aerobically,? Third edition: approved standards, National Committee for Clinical Laboratory Standard Document M7-A3, Villanova, PA.
Neuwinger, H.D., 2000, African traditional medicine: A dictionary of plant use and applications, p. 589, Medpharm Scientific Publishers, Stuttgart, Germany.
Ogundeko, T.O., Idyu, I.I., Ojo, M.T., Idyu, V.C., Ogbole, E.A., Builders, M.I. et al., 2015, ‘Investigation of relative effect of stem-bark extract of Erythrophleum suaveolens on rat phrenic nerve-diaphragm muscle’, Journal of Advances in Medical and Pharmaceutical Sciences 3(1), 24–30. https://doi.org/10.9734/JAMPS/2015/14688
Okoth, D.A., Chenia, H.Y. & Koorbanally, N.A., 2013, ‘Antibacterial and antioxidant activities of flavonoids from Lannea alata (Engl.) Engl. (Anacardiaceae)’, Phytochemistry Letters 6, 476–481. https://doi.org/10.1016/j.phytol.2013.06.003
Ouattara, L., Koudou, J., Zongo, C., Barro, N., Savadogo, A., Bassole, I.H.N. et al., 2011, ‘Antioxidant and antibacterial activities of three species of Lannea from Burkina Faso’, Journal of Applied Sciences 11(11), 157–162.
Prescott, L.M., Harley, J.P. & Klein, D.A., 2002, Microbiology, 6th edn., McGraw-Hill Publishers, pp. 808–823.
Queiroz, E.F., Kuhl, C., Terreaux, C., Mavi, S. & Hostettmann, K., 2003, ‘New dihydroalkylhexenones from Lannea edulis’, Journal of Natural Product 66(4), 578–580. https://doi.org/10.1021/np0202428
Sannigrahi, S., Mazumder, U.K., Pal, D. & Mishra, 2009, ‘Hepatoprotective potential of methanol extract of Clerodendrum infortunatum Linn. against CCl4 induced hepatotoxicity in rats’, Indian Journal of Experimental Biology 5(20), 394–399.
Singleton, V.L., Orthofer, R. & Lamuela-Raventos, R.M., 1999, ‘Analysis of total phenols and other oxidation substrates and anti-oxidation by means of folin-ciocaltev reagent’, Methods in Enzymology 299, 152–178. https://doi.org/10.1016/S0076-6879(99)99017-1
Sofowora, A., 2008, Medicinal plants and traditional medicine in Africa, 3rd edn., Spectrum Books Ltd, Ibadan, Nigeria, p. 439.
Soladoye, M.O., Amusa, N.A., Raji-Esan, S.O., Chukwuma, E.C. & Taiwo, A.A., 2010, ‘Ethnobotanical survey of anti-cancer plants in Ogun-State, Nigeria’, Annals of Biological Research 1(4), 261–273.
Trease, G.C. & Evans, W.C., 1989, Trease & Evan’s textbook of pharmacognosy, 13th edn., Cambridge University press, London, p. 546.
World Health Organization, 2014, Antimicrobial resistance: Global report on surveillance, viewed May 09, 2015.
|

